Are you considering the Watchman device?
Ever since having a TIA/stroke, I certainly have thought a lot about it.
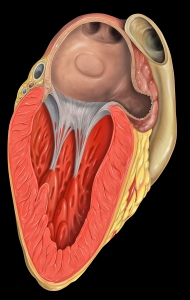
What is it? The Watchman, by Boston Scientific is a little device, sort of like a basket, that can be inserted into the left atrial appendage, theoretically blocking it off and preventing clot formation. As you probably know already, clot formation may lead to Stroke. The device was FDA approved in the US in March, 2015, and has been used in Europe since 2005.
It’s placed in the left atrial appendage via a catheter through an artery in the groin, and if all goes well the patient can discontinue their blood thinner (warfarin, etc.) within six months.
Sounds great, doesn’t it?
I know I’d love to be protected from having another TIA or stroke and not have to take a blood thinner – I’m currently on warfarin + aspirin which makes bicycling, especially mountain biking, quite hazardous. But truthfully, it’s not that I necessarily want to be off the warfarin: I just don’t want to ever have another TIA/stroke. Recall that I had my event while I was already taking Pradaxa (and I never missed a dose). I just want a treatment that is going to work.
But there is some evidence to suggest the Watchman might not be as terrific as it sounds.
A recent study showed that the risk of a major bleed over the course of three years is the same with the Watchman compared to just staying on warfarin. Huh?
This is an excerpt from a Medscape article:
Patients with atrial fibrillation (AF) who received a left atrial appendage closure device (Watchman, Boston Scientific) or stayed on long-term warfarin therapy had similar rates of major bleeding during a mean follow-up of 3.1 years, in pooled analysis of two randomized clinical trials[1]. However, patients who received the device and were able to stop taking warfarin and clopidogrel at 6 months had lower rates of major bleeding from then onward, compared with patients receiving long-term warfarin.
Furthermore, in a very thoughtful, somewhat technical, article CMS Proposal on Watchman Is the Right Decision, Dr John Mandrola, a thought leader in Cardiology and Electrophysiology, agrees with the CMS proposal that “the evidence is sufficient to determine percutaneous left atrial appendage closure therapy using an implanted device is not reasonable and necessary.”
There are two major studies in the US regarding the Watchman. According to Dr Mandrola in the PREVAIL study, “Due to an excess of ischemic strokes, Watchman did not reach noninferiority in this category in the updated analysis presented to the FDA.” In PROTECT-AF study, “ischemic strokes were numerically higher in the Watchman group.” Which, ultimately, “leads one to conclude that the device is not effective.”
As for me, personally, as much as I’d like to believe the Watchman is a solution for me, the evidence, so far, is not convincing. I’m going to wait.
By the way, if any readers have experience with the Watchman PLEASE leave a comment below. We would love to hear from you!
Adverse effects of the Watchman:
“The main adverse events related to this procedure are pericardial effusion, incomplete LAA closure, dislodgement of the device, blood clot formation on the device requiring prolonged oral anticoagulation, and the general risks of catheter-based techniques (such as air embolism). The left atrium anatomy can also preclude use of the device in some patients.”
By the way – I linked a couple of articles from Medscape. I’m not certain but I think you need to be registered for that sight. Sorry.